|
This device, the Stimrouter (Bioness, Valencia, California), obtained Food and Drug Administration (FDA) approval for PNS in the trunk and limbs. The first clinical study to discuss a device strictly designed to stimulate a peripheral nerve was published by Deer and colleagues as an Investigational Device Exemption (IDE) study on peripheral nerve pain (Deer et al., 2016). Unfortunately, this potential landmark study failed to meet the primary end point and brought attention to major adverse events such as lead erosion and lead migration (Saper et al., 2011). Eventually, a randomized multicenter prospective study was performed to access efficacy and safety of PNS for the migraine indication (Saper et al., 2011). This procedure fundamentally changed in 1999, when Weiner described using percutaneous leads originally designed for the spinal cord to treat occipital nerve-induced headaches (Weiner & Reed, 1999). This approach was modified and involved placing a transposed fascial graft between the nerve and lead, though fell out of favor due to the complexity of the surgery and adverse effects (Law et al., 1980). The original approach described by Wall and Sweet was an open approach where the surgeon dissected the tissue to visualize the nerve and apply the lead (Long, 1977). PNS was first used in Philadelphia, Pennsylvania 60 years ago to treat pain in the head and neck (Shelden, 1966). PNS is considered a low risk procedure, but also has a lower threshold for efficacy, based on limited high evidence studies (Deer et al., 2016). These principles include understanding the usefulness of a trial prior to an implant, optimal stimulation parameters, appropriate image guidance for optimal lead placement, and specific neural targets which have evidence for success. Thus, in this review we discuss the principles that govern successful outcomes for PNS. However, for PNS to be successful, understanding of outcomes is not sufficient. If patients do not significantly improve with conservative therapies, PNS is a viable nerve treatment option to treat pain secondary to peripheral nerve injury (Van Buyten et al., 2015 Nagel et al., 2014 Pereira & Aziz, 2014). thermal or chemical) and neurostimulation (Bates et al., 2019). Treatment options for peripheral nerve injury include: physical therapy, oral medications, ablative therapies (e.g. The interventional pain physician may also do diagnostic peripheral nerve blocks using local anesthetic(s) to aid in diagnosis (Bates et al., 2019). Imaging, such as MRI or ultrasound along with diagnostic tests, such as electromyelogram or nerve conduction studies are often used to aid in diagnosis (Rangavajla et al., 2014 Aminoff, 2004). The diagnosis is made by pain in the distribution of the peripheral nerve, based on both history and examination. 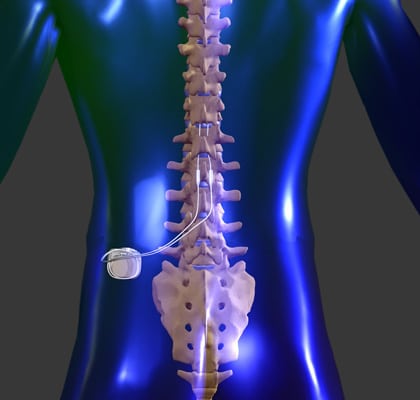 
It is estimated that up to 10 % of those impacted by chronic pain may have an origin related to peripheral nerve pathology (Breivik et al., 2006). These problems can lead to suffering and disability, which is a major economic burden to both the patient and the healthcare community (Breivik et al., 2006 Duenas et al., 2016). Once chronic pain has developed, the patient may experience allodynia, hyperalgesia, and loss of movement or function (Bennett & Xie, 1988). 
Common examples of chronic pain caused by peripheral nerve injury include ilioinguinal and/or iliohypogastric nerve pain after inguinal herniorrhaphy, sural nerve injury after podiatric surgery leading to foot pain, intercostal nerve pain after thoracotomy, and facial pain after ophthalmic infection of herpes zoster (Alfieri et al., 2006 Gerner, 2008 Primadi et al., 2016 Opstelten & Zaal, 2005). These cell bodies serve as first pass junctions for the transmitted signal, and may lead to a hyperexcitable state with resultant changes in the area of the peripheral nerve, the peripheral ganglia, the spinal cord, and at the level of the anterior cingulate cortex (Tajerian et al., 2013). Peripheral changes in chemical mediators can lead to pathological nerve firing, triggering changes in the cell bodies of somatosensory neurons located in the peripheral ganglia (dorsal root or trigeminal ganglia). However, persistence in nociception or peripheral nerve dysfunction can lead to the development of chronic pain which can have profound consequences to that organism and its social structure (Campbell, 2008 Costigan et al., 2009). In most situations after mechanical or metabolic trauma, transduction and nociception can be beneficial, informing an organism to retreat or protect. 
Touch, proprioception, temperature, and nociception influence our perception of the world (Benarroch et al., 2018). The peripheral nervous system is an integral part of the body’s communication with the environment (Kiernan et al., 2013).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
